Really interesting read. Thanks - and to all the contributors to the conversation too !
Navigation
Install the app
How to install the app on iOS
Follow along with the video below to see how to install our site as a web app on your home screen.
Note: This feature may not be available in some browsers.
More options
Style variation
Welcome to ScapeCrunch
We are ScapeCrunch, the place where planted aquarium hobbyists come to build relationships and support each other. When you're tired of doom scrolling, you've found your home here.
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Journal Recovering from disaster: My 120P journey.
- Thread starter bradquade
- Start date
- Featured
- Tagged users None
Hello, of course I check your thread every day to see if there are any updates 
I have suffered a lot from this algae. Just like you, there is no method I haven’t tried. Chemicals definitely do not work. It’s not related to the substrate either, because I reset the tank many times. I even bought brand new soil
This is definitely caused by a relationship between iron–phosphate or iron–micro–phosphate. (It appears especially when N and P drop to zero while free iron remains high; afterward phosphate feeds it.)
When this algae appears, it is always clear that there is an iron-related issue in the plants. I have tested this many times with photometer devices. Either iron is depleted while phosphate is high, or phosphate is zero while iron is high.
For example, if I increase potassium even by 1 ppm, the algae increases by 50% the next day. Potassium definitely “unlocks” something. But if I dose 0.5 ppm daily, the algae stays stable but does not die.
EDDHA iron increased the algae dramatically — a 0.1 ppm dose caused a 50% increase in just one day. With EDTA iron, I get the usual white tips (new growth issues). DTPA iron keeps the algae stable. Interestingly, Fe gluconate makes plants look great — you can immediately see color improvement — but of course the algae also grows very well.
It feels like an endless cycle.
Because my pH and CO₂ are very high, I previously avoided adding livestock to my tanks, since they would die and it bothered me. But now I wanted to test whether they are really effective.
First, I bought 500 mixed Neocaridina shrimp. In my first tank, I set KH 1 and GH 10. The pH is 6.1 when CO₂ is on. I added 100 shrimp to this tank. They didn’t have much effect on the algae because it was already too heavy. I tested them for about 1 month.
My second tank is GH 1, KH 0, TDS 30, pH 5.3. I added another 100 shrimp there. This tank had the most algae — I was removing it by the cup. It was insane.
My third tank is one of the most problematic ones. I had this same algae for years. I couldn’t adjust the light intensity, and it was very strong. I can say I never saw it algae-free
Then I asked AI: “Tell me everything about this algae — how it lives, what kills it, what eats it, etc.” We had a long discussion It suggested things like barley water and salicylic acid (this actually kills it).
It suggested things like barley water and salicylic acid (this actually kills it).
Then I asked: which organism eats it the most? It said SAE (Crossocheilus siamensis).
So I bought 50 from a wholesaler. I added:
At first they were quite stressed. Then I added oxygen at night. In the morning, 6 hours before lights on, I stopped oxygen and started CO₂. They adapted very well.
In all tanks, the algae dropped to zero. It took about 5–10 days.
But here’s the interesting part: the algae is still forming. Because my filter is a side sump and livestock cannot enter it, algae continues to grow there.
So the water chemistry is not fixed. It’s just that whenever algae appears on plants, the SAEs eat it immediately.
If I had tried this earlier, I wouldn’t have spent so much effort. If you cannot solve this problem, I strongly recommend SAE — but 3–4 fish is not enough. In groups, they work extremely well.
I’m sending a sample photo of my second tank (sump algae and plants). Colors haven’t fully settled yet, but it seems to be improving.
By the way, I didn’t change the fertilization at all. I’m still dosing 0.4 ppm K
(“Of course, this is a video of its cleaned state.”)
“I mean, my friend, they are literally tearing it apart and eating it right in front of my eyes.”
“I think the algae I had was this one — Rhizoclonium. If I leave it for a long time, it turns into this form. It has a slimy texture. It’s green and soft, and it breaks apart easily. But once it attaches to the leaves, it becomes difficult to remove.”

“SAE in action.”

“Algae growth in the sump area.”

“And as a result, once the algae died, the plants also started to recover. It seems like the algae was rapidly consuming the nutrients as well.”


“I hope this experience I had can be helpful to you. I’m still following the thread — please keep us updated from time to time.”
I have suffered a lot from this algae. Just like you, there is no method I haven’t tried. Chemicals definitely do not work. It’s not related to the substrate either, because I reset the tank many times. I even bought brand new soil
This is definitely caused by a relationship between iron–phosphate or iron–micro–phosphate. (It appears especially when N and P drop to zero while free iron remains high; afterward phosphate feeds it.)
When this algae appears, it is always clear that there is an iron-related issue in the plants. I have tested this many times with photometer devices. Either iron is depleted while phosphate is high, or phosphate is zero while iron is high.
For example, if I increase potassium even by 1 ppm, the algae increases by 50% the next day. Potassium definitely “unlocks” something. But if I dose 0.5 ppm daily, the algae stays stable but does not die.
EDDHA iron increased the algae dramatically — a 0.1 ppm dose caused a 50% increase in just one day. With EDTA iron, I get the usual white tips (new growth issues). DTPA iron keeps the algae stable. Interestingly, Fe gluconate makes plants look great — you can immediately see color improvement — but of course the algae also grows very well.
It feels like an endless cycle.
Because my pH and CO₂ are very high, I previously avoided adding livestock to my tanks, since they would die and it bothered me. But now I wanted to test whether they are really effective.
First, I bought 500 mixed Neocaridina shrimp. In my first tank, I set KH 1 and GH 10. The pH is 6.1 when CO₂ is on. I added 100 shrimp to this tank. They didn’t have much effect on the algae because it was already too heavy. I tested them for about 1 month.
My second tank is GH 1, KH 0, TDS 30, pH 5.3. I added another 100 shrimp there. This tank had the most algae — I was removing it by the cup. It was insane.
My third tank is one of the most problematic ones. I had this same algae for years. I couldn’t adjust the light intensity, and it was very strong. I can say I never saw it algae-free
Then I asked AI: “Tell me everything about this algae — how it lives, what kills it, what eats it, etc.” We had a long discussion
Then I asked: which organism eats it the most? It said SAE (Crossocheilus siamensis).
So I bought 50 from a wholesaler. I added:
- 5 to tank 1
- 25 to tank 2
- 20 to tank 3
At first they were quite stressed. Then I added oxygen at night. In the morning, 6 hours before lights on, I stopped oxygen and started CO₂. They adapted very well.
In all tanks, the algae dropped to zero. It took about 5–10 days.
But here’s the interesting part: the algae is still forming. Because my filter is a side sump and livestock cannot enter it, algae continues to grow there.
So the water chemistry is not fixed. It’s just that whenever algae appears on plants, the SAEs eat it immediately.
If I had tried this earlier, I wouldn’t have spent so much effort. If you cannot solve this problem, I strongly recommend SAE — but 3–4 fish is not enough. In groups, they work extremely well.
I’m sending a sample photo of my second tank (sump algae and plants). Colors haven’t fully settled yet, but it seems to be improving.
By the way, I didn’t change the fertilization at all. I’m still dosing 0.4 ppm K
(“Of course, this is a video of its cleaned state.”)
“I mean, my friend, they are literally tearing it apart and eating it right in front of my eyes.”
“I think the algae I had was this one — Rhizoclonium. If I leave it for a long time, it turns into this form. It has a slimy texture. It’s green and soft, and it breaks apart easily. But once it attaches to the leaves, it becomes difficult to remove.”

“SAE in action.”

“Algae growth in the sump area.”

“And as a result, once the algae died, the plants also started to recover. It seems like the algae was rapidly consuming the nutrients as well.”


“I hope this experience I had can be helpful to you. I’m still following the thread — please keep us updated from time to time.”
Attachments
- Thread starter
- #103
@mrtank50 Thanks for sending this update! Your tank is looking great! It seems like the fish really helped you overcome the issues. I'm always wary of adding animals to my tanks because I'm not very careful with CO2, but will consider this if I end up throwing in the towel. I'm inclined to agree with you that the algae is related to iron availability and will demonstrate that below.
I haven't been updating because I've continued being plagued by completely white/bleached growth on many of my plants as shown in some of the previous updates. I thought waiting it out and letting the plants adapt was the right call, but something very strange happened that made me realize there was, once again, something wrong. The Limnophila wilsonii side shoots eventually emerged from the bottom growth and as soon as they were exposed to higher light they turned white and the growing tips died. Clearly adaptation wasn't making a difference, so I started experimenting by moving plants between my 10 gallons that behave well and the 120P that behaves poorly. Below is a summary of the differences between the two tanks that I was experimenting with just for reference.
Experiment 1: Floaters from 10g to 120P
I had a ton of red root floaters in the 10 g. I figured I could figure out CO2 issues vs nutrient issues by seeing what new growth on the floaters looked like. I added the floaters to the 120P and they grew normal green growth. That implies that nutrient availability is not the issue and was kind of a bummer because that's an easy fix. When I took the floaters out to throw them away, I noticed that all of the roots died even though they were growing still. The roots were completely black and melted and that's weird because root growth has been a major issue in the tank for months.
Experiment 2: Glosso from 10g to 120P
The 10 gallon has a carpet of Glossostigma and it grows like crazy. I often have to trim it and because I have floaters, I sometimes leave pieces of Glosso floating in the tank. One piece was floating for a couple weeks and instead of throwing it away, I moved it to the 120P. It was floating and growing well in the 10g, so it should be adapted to getting nutrients from the water column. Three days after moving it, it turned white on the growing tip (shown below) and the growing tip died. That's weird because that is what's happening to the other plants in the tank since I changed the substrate.

Experiment 3: Limnophila heterophylla from 10g to 120P
I trimmed the Limno. heterophylla from the 10 gallon and instead of throwing the trimmings away I moved a couple stems to the 120P. Three days later it was completely white on the growing tip. Notice a trend here? At this point I checked the CO2 and the pH drop was still 1.5 and both drop checkers were lime green to yellow at lights on. CO2 cannot be the issue here.
Experiment 4: Hygrophila triflora from 120P to 10g
This plant was not happy with the substrate change in the 120P. Every single stem stunted super bad and stopped growing (shown below). I pulled up a few of the stems and they had grown 0 roots despite being planted for a few weeks. That was concerning so I took a stem, moved it to the 10 gallon, and left it floating on the surface of the tank. Five or six days later when I was removing floaters I took out the stem. It had 2" of root growth and side shoots growing at every node. That told me that something in the 120P needs to change to match the 10 gallon.

What should I change?
The obvious answer here is that I should change the substrate to Fluval Stratum. I don't want to do this though. I want to figure out how to grow plants in inert substrate again since its easy once conditions are nailed down. I also figured that the substrate itself isn't the answer because the Hygrophila stem was floating and never actually experienced the substrate. What can active substrate influence outside of water column nutrients that is also very different between the two tanks? pH was the next obvious answer. I never cared about pH because there are people keeping very successful planted tanks at a huge variety of pHs. I had never had a successful tank running at less than 5.8-5.9 though, so I figured it was worth changing. During my next water change I added 1 dKH from potassium bicarbonate, which brought the pH up to ~5.5 at max CO2.
Experiment 1 at pH 5.5: Glosso and red root floaters from the 10g to 120P
Since Glosso responded so quickly to the transition between tanks, I used this as the control plant to see if pH made a difference in the plant whitening. I added a new stem, let it sit for a week, and it was a little pale, but not completely white (shown below). Nutrient dosing and CO2 had not changed at all, so this phenomenon was driven entirely be the increased pH/KH. When I added the Glosso I also added red root floaters again. This time the roots didn't die. It was very interesting seeing that the pH made a difference in how the plants responded to the transition. I was also looking at the glass one day ~1 week after increasing the pH and noticed a ton of copepods. That was also encouraging because I haven't seen any tiny creatures in the tank for a long, long time. The pH was still much lower than 10g though, so I decided to up the KH to 2.

Observations from KH2:
For reference, I added 1 dKH around 3/13 and then upped it to 2dKH around 3/20. At 2dKH, the pH in the tank reaches 5.8 at maximum CO2. When I measure before CO2 comes on, it's typically pH 6.2-6.4. This is because I have little surface agitation and my tank doesn't lose much CO2 throughout the night. These pH values are completely in line with my tanks that were very successful back in Texas.

Limnophila wilsonii immediately recovered after increasing the KH. The bottoms look super ugly, but I can see many little green tops growing in. The change from 3/21 to 3/24 is very dramatic.

I moved the Hygrophila triflora stem that recovered in the 10g back into the 120P. In the last 4 days, the roots have grown a huge amount. This is very interesting to see because this plant didn't root at all during the time it was in the tank before I moved it to the 10g. You can also see that the new growth is fairly normal looking compared to the previous picture.

This is the Limnophilla heterophylla I moved into the 120P from the 10g. Notice how it's green, white, and then green again. The original green is from the 10g. The white is from the 120P when pH was 5. The green is from the 120P when pH was ~5.8.

I was really happy to see that the Rotala macrandra 'Pink' was putting out aerial roots on March 9th, but ever since I upped the pH the aerial root growth has been crazy. It's not just this plant either. Almost every plant in the tank is growing aerial roots at a rate that I consider normal and that has not happened in this tank for a long time.

Here is the tank as a couple days ago. The plants that were growing well before adding KH are growing extremely fast now. The plants that turned completely white are growing side shoots and appear to be in the early stages of recovery. Some of them are already turning green on the growing tips, like Limnophila willsonii, and others are just starting to put out roots. Recovery is faster in some plants than others and I hope the improvements in the new growth continue. I really want to increase the light intensity so I can get the color back in the plants, but it's still too early for that.
Why does the pH matter?
pH is a logarithmic scale and the concentration of protons in the water increases 10x when you go from pH 6 to pH 5. When the concentration of protons in the water gets too high, they can prevent plants from taking up nutrients correctly. My plants were exhibiting classical symptoms of calcium deficiency, which was literally impossible given the amount of calcium in the water. It turns out that high proton concentrations directly antagonize Ca/Mg/K binding in roots, which would cause pH-induced deficiency. Iron is a bit weird and unlike other nutrients, becomes more soluble as the pH decreases. The issue is that as availability increases, it can become toxic. Many people, including myself, ignore pH, but it's worth thinking about if you're having long term issues.
I haven't been updating because I've continued being plagued by completely white/bleached growth on many of my plants as shown in some of the previous updates. I thought waiting it out and letting the plants adapt was the right call, but something very strange happened that made me realize there was, once again, something wrong. The Limnophila wilsonii side shoots eventually emerged from the bottom growth and as soon as they were exposed to higher light they turned white and the growing tips died. Clearly adaptation wasn't making a difference, so I started experimenting by moving plants between my 10 gallons that behave well and the 120P that behaves poorly. Below is a summary of the differences between the two tanks that I was experimenting with just for reference.
| 10 g tank | 120P | |
| Substrate | Fluval Stratum | Blasting Sand |
| Water source | RO water | RO water |
| Water change | Biweekly 70% | Weekly 70% |
| Ca/Mg | 30/10 | 30/10 |
| KH | 0 | 0 |
| NPK | 2 EI doses front loaded | 2 EI doses front loaded and 1x mid week |
| Traces | 0.3 PPM Fe front loaded | 0.45 PPM Fe in 3 doses |
| CO2 | 0.8-0.9 pH drop | Drop checkers in front two corners are yellow at lights on |
| pH | 6.3 at max CO2 | 5.0 at max CO2 |
Experiment 1: Floaters from 10g to 120P
I had a ton of red root floaters in the 10 g. I figured I could figure out CO2 issues vs nutrient issues by seeing what new growth on the floaters looked like. I added the floaters to the 120P and they grew normal green growth. That implies that nutrient availability is not the issue and was kind of a bummer because that's an easy fix. When I took the floaters out to throw them away, I noticed that all of the roots died even though they were growing still. The roots were completely black and melted and that's weird because root growth has been a major issue in the tank for months.
Experiment 2: Glosso from 10g to 120P
The 10 gallon has a carpet of Glossostigma and it grows like crazy. I often have to trim it and because I have floaters, I sometimes leave pieces of Glosso floating in the tank. One piece was floating for a couple weeks and instead of throwing it away, I moved it to the 120P. It was floating and growing well in the 10g, so it should be adapted to getting nutrients from the water column. Three days after moving it, it turned white on the growing tip (shown below) and the growing tip died. That's weird because that is what's happening to the other plants in the tank since I changed the substrate.

Experiment 3: Limnophila heterophylla from 10g to 120P
I trimmed the Limno. heterophylla from the 10 gallon and instead of throwing the trimmings away I moved a couple stems to the 120P. Three days later it was completely white on the growing tip. Notice a trend here? At this point I checked the CO2 and the pH drop was still 1.5 and both drop checkers were lime green to yellow at lights on. CO2 cannot be the issue here.
Experiment 4: Hygrophila triflora from 120P to 10g
This plant was not happy with the substrate change in the 120P. Every single stem stunted super bad and stopped growing (shown below). I pulled up a few of the stems and they had grown 0 roots despite being planted for a few weeks. That was concerning so I took a stem, moved it to the 10 gallon, and left it floating on the surface of the tank. Five or six days later when I was removing floaters I took out the stem. It had 2" of root growth and side shoots growing at every node. That told me that something in the 120P needs to change to match the 10 gallon.

What should I change?
The obvious answer here is that I should change the substrate to Fluval Stratum. I don't want to do this though. I want to figure out how to grow plants in inert substrate again since its easy once conditions are nailed down. I also figured that the substrate itself isn't the answer because the Hygrophila stem was floating and never actually experienced the substrate. What can active substrate influence outside of water column nutrients that is also very different between the two tanks? pH was the next obvious answer. I never cared about pH because there are people keeping very successful planted tanks at a huge variety of pHs. I had never had a successful tank running at less than 5.8-5.9 though, so I figured it was worth changing. During my next water change I added 1 dKH from potassium bicarbonate, which brought the pH up to ~5.5 at max CO2.
Experiment 1 at pH 5.5: Glosso and red root floaters from the 10g to 120P
Since Glosso responded so quickly to the transition between tanks, I used this as the control plant to see if pH made a difference in the plant whitening. I added a new stem, let it sit for a week, and it was a little pale, but not completely white (shown below). Nutrient dosing and CO2 had not changed at all, so this phenomenon was driven entirely be the increased pH/KH. When I added the Glosso I also added red root floaters again. This time the roots didn't die. It was very interesting seeing that the pH made a difference in how the plants responded to the transition. I was also looking at the glass one day ~1 week after increasing the pH and noticed a ton of copepods. That was also encouraging because I haven't seen any tiny creatures in the tank for a long, long time. The pH was still much lower than 10g though, so I decided to up the KH to 2.

Observations from KH2:
For reference, I added 1 dKH around 3/13 and then upped it to 2dKH around 3/20. At 2dKH, the pH in the tank reaches 5.8 at maximum CO2. When I measure before CO2 comes on, it's typically pH 6.2-6.4. This is because I have little surface agitation and my tank doesn't lose much CO2 throughout the night. These pH values are completely in line with my tanks that were very successful back in Texas.

Limnophila wilsonii immediately recovered after increasing the KH. The bottoms look super ugly, but I can see many little green tops growing in. The change from 3/21 to 3/24 is very dramatic.

I moved the Hygrophila triflora stem that recovered in the 10g back into the 120P. In the last 4 days, the roots have grown a huge amount. This is very interesting to see because this plant didn't root at all during the time it was in the tank before I moved it to the 10g. You can also see that the new growth is fairly normal looking compared to the previous picture.

This is the Limnophilla heterophylla I moved into the 120P from the 10g. Notice how it's green, white, and then green again. The original green is from the 10g. The white is from the 120P when pH was 5. The green is from the 120P when pH was ~5.8.

I was really happy to see that the Rotala macrandra 'Pink' was putting out aerial roots on March 9th, but ever since I upped the pH the aerial root growth has been crazy. It's not just this plant either. Almost every plant in the tank is growing aerial roots at a rate that I consider normal and that has not happened in this tank for a long time.

Here is the tank as a couple days ago. The plants that were growing well before adding KH are growing extremely fast now. The plants that turned completely white are growing side shoots and appear to be in the early stages of recovery. Some of them are already turning green on the growing tips, like Limnophila willsonii, and others are just starting to put out roots. Recovery is faster in some plants than others and I hope the improvements in the new growth continue. I really want to increase the light intensity so I can get the color back in the plants, but it's still too early for that.
Why does the pH matter?
pH is a logarithmic scale and the concentration of protons in the water increases 10x when you go from pH 6 to pH 5. When the concentration of protons in the water gets too high, they can prevent plants from taking up nutrients correctly. My plants were exhibiting classical symptoms of calcium deficiency, which was literally impossible given the amount of calcium in the water. It turns out that high proton concentrations directly antagonize Ca/Mg/K binding in roots, which would cause pH-induced deficiency. Iron is a bit weird and unlike other nutrients, becomes more soluble as the pH decreases. The issue is that as availability increases, it can become toxic. Many people, including myself, ignore pH, but it's worth thinking about if you're having long term issues.
What's pH at in the 120 right before CO2 turns on in the morning? Maybe more aeration to raise the pH and oxygen levels at night might be worth a try.
Limnophila wilsonii immediately recovered after increasing the KH
FWIW, Limnophila wilsonii is growing like bananas for me
CO2 tests at 15 ppm, so that the pH only drops to about 7.4 at peak CO2.
Last edited:
I was just thinking about this journal, glad to see an update!
I also find it interesting that we're on similar paths. I recently started raising my KH of my incoming fresh RO water from 0 to ~1.6 via KHCO3, too, and also saw some great improvements. I ALSO had some pale growth tips that corrected quickly when increasing KH.
Is there any chance that this improvement in your tank could also be from the extra K added? I have it memorized that adding ~1dKH via KHCO3 raises the K by ~14ppm, too:
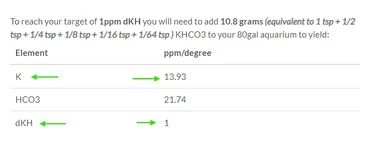
In my tank, I'm using KHCO3 to replace K2SO4 for the "extra" K needed after WC, since my Macros solution doesn't provide enough. So my K levels stayed about the same when I started raising my KH, but is that the case for yours, too?
Was this just an increase in KH with similar K levels, or did you also benefit from the increase in K (between 14ppm and 28ppm across 1-2dKH)?
During my next water change I added 1 dKH from potassium bicarbonate, which brought the pH up to ~5.5 at max CO2.
The pH was still much lower than 10g though, so I decided to up the KH to 2.
I also find it interesting that we're on similar paths. I recently started raising my KH of my incoming fresh RO water from 0 to ~1.6 via KHCO3, too, and also saw some great improvements. I ALSO had some pale growth tips that corrected quickly when increasing KH.
Is there any chance that this improvement in your tank could also be from the extra K added? I have it memorized that adding ~1dKH via KHCO3 raises the K by ~14ppm, too:

In my tank, I'm using KHCO3 to replace K2SO4 for the "extra" K needed after WC, since my Macros solution doesn't provide enough. So my K levels stayed about the same when I started raising my KH, but is that the case for yours, too?
Was this just an increase in KH with similar K levels, or did you also benefit from the increase in K (between 14ppm and 28ppm across 1-2dKH)?
This is my guess. I've been growing Limnophila wilsonii in zero dKH for quite a while. Have never seen anything like white tips ever. Grows like a weed with big green fluffy tops. Branches like mad. I toss loads out as if left unchecked it can become huge.In my tank, I'm using KHCO3 to replace K2SO4 for the "extra" K needed after WC, since my Macros solution doesn't provide enough. So my K levels stayed about the same when I started raising my KH, but is that the case for yours, too?
Was this just an increase in KH with similar K levels, or did you also benefit from the increase in K (between 14ppm and 28ppm across 1-2dKH)?
My guess is that he's seeing the effect of more K in the water column.
- Thread starter
- #108
@TRyan The CO2 will turn on in 2 hours and the pH is currently 6.2. It hits ~5.8 when the CO2 concentration is at max. I figured increasing the pH with a little bit of KH would be easier than degassing the CO2 and reoptimizing injection.
@Naturescapes_Rocco When I changed the substrate in the tank I was dosing 34PPM N03-10 PPM P04-44 PPM K and 0.45 PPM Fe from Burr micros weekly. This is what the tank was maintained on when all of the plants turned white, roots didn't grow, and many of the tops started melting. When I started pulling plants up and saw zero root growth over a ~3-4 week period, I changed to my classic dry dosing strategy that adds 30 PPM NO3-7 PPM PO4-32 PPM K. This was maintained for ~1 week before increasing the KH with potassium bicarbonate and once I added potassium bicarbonate, I stopped adding K2SO4. Now my dosing is roughly 30 PPM NO3-7 PPM PO4-40 PPM K weekly with the same Fe+micros. This is slightly lower N, P, and K than when the white growth began, so I'm inclined to think the change is related to the bicarbonate addition more than potassium.
It's also worth mentioning that plants recovered fairly quickly when moved from the 120P to the 10g tank and that tank is dosed with 17 PPM NO3-4 PPM PO4-19 PPM K. If the plants were just hungry for K, why would they recover so quickly in conditions half as rich?
@GreggZ My weekly K dosing is 4 PPM lower than when the white growth became an issue, so it's definitely not from a larger amount of K in the water column.
@Naturescapes_Rocco When I changed the substrate in the tank I was dosing 34PPM N03-10 PPM P04-44 PPM K and 0.45 PPM Fe from Burr micros weekly. This is what the tank was maintained on when all of the plants turned white, roots didn't grow, and many of the tops started melting. When I started pulling plants up and saw zero root growth over a ~3-4 week period, I changed to my classic dry dosing strategy that adds 30 PPM NO3-7 PPM PO4-32 PPM K. This was maintained for ~1 week before increasing the KH with potassium bicarbonate and once I added potassium bicarbonate, I stopped adding K2SO4. Now my dosing is roughly 30 PPM NO3-7 PPM PO4-40 PPM K weekly with the same Fe+micros. This is slightly lower N, P, and K than when the white growth began, so I'm inclined to think the change is related to the bicarbonate addition more than potassium.
It's also worth mentioning that plants recovered fairly quickly when moved from the 120P to the 10g tank and that tank is dosed with 17 PPM NO3-4 PPM PO4-19 PPM K. If the plants were just hungry for K, why would they recover so quickly in conditions half as rich?
@GreggZ My weekly K dosing is 4 PPM lower than when the white growth became an issue, so it's definitely not from a larger amount of K in the water column.
That's interesting. So it's not the K. But I am still doubtful it has anything to with dKH . Of course I have been wrong before but I am just skeptical based on my own experience and the experience of others that I know in the hobby.@GreggZ My weekly K dosing is 4 PPM lower than when the white growth became an issue, so it's definitely not from a larger amount of K in the water column.
Keep in mind with this hobby it's difficult to draw accurate conclusions about cause/effect. There are lots of variables at play in a tank. If low dKH caused stunting with white tips at low/zero dKH I would expect to see it my tank and others but i don't.
And it's not just the wilsonii. You mention Rotala Macranda as well. I've been keeping several Macranda's for many, many years in zero dKH conditions and so have lots and lots of other people (i.e. Dennis Wong).
You need to look at this way sometimes. If dKH is truly the issue then others should be able to induce similar stunting by putting them in low/no dKH environments. I know too many people with very low to no dKH tanks where the same plants are thriving.
And please don't take this as argumentative. It's not. I can see you had some pretty serious stunting and have been frustrated.
Have you considered the tank might just be finally coming into balance/stability after the substrate swap? It's not been that long and a bio field takes some time mature after a disruption like that. To me that is much more likely than the dKH.
Either way yours is an interesting case. Curious to hear what others think.
I agree with this:
Recently I, too, had some white tips (especially on my new Syngonanthus species) and GSA occur when I over-dosed my Macros. Even with 70%+ water changes, I had two weeks where my PO4 was reaching levels of 12-14ppm (tested with Hanna High-range PO4 checker AND salifert PO4 (diluted)). What I noticed is that when my macros got too high, I saw white tips and stunted growth.
My guess is that, especially at low pH, the concentrated phosphate was perhaps binding, or preventing, the uptake of something? Or, some micros weren't getting into my plant tissues because of excess K? I tried dosing more micros (reaching 0.7ppm Fe as proxy per week) but it didn't help, and caused other potential issues.
Who knows what one thing DID work, but I did two things that seems to have fixed it:
Raised dKH with KHCO3, and ultimately lowered the macros dose, as well as the residual macros level in the water column with a few extra bi-weekly water changes.
I went from ~36/12/50 NO3/PO4/K weekly dosing, back to 30/9/39 total weekly dosing (all with 70% weekly water changes). I also did multiple water change cycles to help remove the excess macros and micros from the tank.
Stunted white tips went away in about 2 days. Plants look green again and pearling is back to max level.
My thoughts are, maybe the same happened for you @bradquade ?
Reducing my macros to more sane levels immediately fixed the stunting issues, maybe it did for you, too.
Either way, I am genuinely thrilled that you're on the track to healthy plants again. I know the frustration of battling issues for months on end, wanting to either give up or finally solve the puzzle (or die trying). I hope this change in your tank has brought the positivity back, it definitely looks like a promising improvement!
Keep in mind with this hobby it's difficult to draw accurate conclusions about cause/effect. There are lots of variables at play in a tank.
Recently I, too, had some white tips (especially on my new Syngonanthus species) and GSA occur when I over-dosed my Macros. Even with 70%+ water changes, I had two weeks where my PO4 was reaching levels of 12-14ppm (tested with Hanna High-range PO4 checker AND salifert PO4 (diluted)). What I noticed is that when my macros got too high, I saw white tips and stunted growth.
My guess is that, especially at low pH, the concentrated phosphate was perhaps binding, or preventing, the uptake of something? Or, some micros weren't getting into my plant tissues because of excess K? I tried dosing more micros (reaching 0.7ppm Fe as proxy per week) but it didn't help, and caused other potential issues.
Who knows what one thing DID work, but I did two things that seems to have fixed it:
Raised dKH with KHCO3, and ultimately lowered the macros dose, as well as the residual macros level in the water column with a few extra bi-weekly water changes.
I went from ~36/12/50 NO3/PO4/K weekly dosing, back to 30/9/39 total weekly dosing (all with 70% weekly water changes). I also did multiple water change cycles to help remove the excess macros and micros from the tank.
Stunted white tips went away in about 2 days. Plants look green again and pearling is back to max level.
My thoughts are, maybe the same happened for you @bradquade ?
Similar to what I was dosing initially. And if I remember, you perform slightly smaller water change %, right? so your accumulation could have been even higher than mine. Perhaps your PO4 reached levels of 12-14ppm, or maybe even more? My gut feeling is that much PO4/K causes serious issues in our plant systems regarding micros consumption, leading to stunting (with low color, versus stunting with burnt color).@Naturescapes_Rocco When I changed the substrate in the tank I was dosing 34PPM N03-10 PPM P04-44 PPM K and 0.45 PPM Fe from Burr micros weekly. This is what the tank was maintained on when all of the plants turned white, roots didn't grow, and many of the tops started melting.
It could be the bicarbonate, but also this is a non-negligible decrease in macros, too! Especially if you did some extra water changes to balance things out. Perhaps the levels of macros are now in a zone where they aren't preventing something from happening with your plants? The reason I think that is that is SO similar to what happened with mine. It was clear that with low pH and HIGH macros, I got white tips and stunting despite dosing solid micros.Now my dosing is roughly 30 PPM NO3-7 PPM PO4-40 PPM K weekly with the same Fe+micros. This is slightly lower N, P, and K than when the white growth began, so I'm inclined to think the change is related to the bicarbonate addition more than potassium.
Reducing my macros to more sane levels immediately fixed the stunting issues, maybe it did for you, too.
Either way, I am genuinely thrilled that you're on the track to healthy plants again. I know the frustration of battling issues for months on end, wanting to either give up or finally solve the puzzle (or die trying). I hope this change in your tank has brought the positivity back, it definitely looks like a promising improvement!
Last edited:
This is a great example of why it's hard to pinpoint cause/effect. In this case you really have three changes, raised dKH, lowered macros, and bi weekly water changes.Who knows what one thing DID work, but I did two things that seems to have fixed it:
Raised dKH with KHCO3, and ultimately lowered the macros dose, as well as the residual macros level in the water column with bi-weekly water changes.
Whenever you change more than one parameter at a time (which most people do) it's hard to say which really made a difference. Was it really raising dKH? If you lowered it now would the same problems creep up? Same for dosing, if you raised dosing now would it induce the same symptoms? And bi weekly water changes are well known to whip a tank into shape. Could it be that was the one that mattered? Or was it all three?
Who knows. Either way glad to see it's working better for you. When you have time you should alter ONLY one at time just to see what happens. I've done this many times over the years and can be eye opening.
- Thread starter
- #112
@GreggZ I don't take your perspective as an argument. I think its completely reasonable to look at other peoples systems and use their experiences to establish a baseline. Focusing on success at 0 dKH ignores a key part of what I'm thinking made a difference. 0 dKH is not inherently bad and I have multiple tanks running successfully with 0 dKH. I think the lack of buffering capacity in the 120P is driving the pH to a level that it is so low that it inhibits uptake in certain plant species. One of my 0 dKH tanks has a pH of 6.8 because it has no CO2. Another has a pH of 6.2 at max CO2 because it has soil substrate. The 120P with no soil, no buffering capacity, and high CO2 spent the majority of its time at pH 5. This is fairly low and is outside of the range that Dennis recommends on 2 hr aquarist. He writes "a pH of 6 to 7 - slightly acidic, is what most planted tanks function well optimally at, and this is also why all aquasoils contain peat which slightly acidifies the tank and lowers KH levels."
I know there are people out there that have successful tanks with pH in the high 4 to low 5 range and I can't explain why my tank would behave differently. There are too many variables when you account for nutrient dosing levels, fauna, CO2 levels, substrate, etc. All I know is that increasing the pH in the tank to match my other tanks, both current and historical, made a positive difference and put it in line with ideal pH targeted by aquasoil manufacturers.
If the tank continues progressing in a positive manner and I have a tank of fully healthy plants, I'll probably do the experiment where I remove the KHCO3 and see if I get stunting/white growth. Single variable experiments are not productive at this moment though because the tank is in rough condition.
@Naturescapes_Rocco I've been doing 70% water changes for a while. I struggled to consistently do 50% because I always remove more water than I want when I'm cleaning up the tank. Since I'm thinking the issue is pH, I'm wondering what yours was at max CO2 with 0dKH vs now. I'm wondering if yours was as low as mine.
It's also possible that a slightly decrease in macros made a difference. I try not to think too hard about these things and just go off of what works. Lower macros+higher KH seems to make a difference so I'm sticking with it for some time.
I know there are people out there that have successful tanks with pH in the high 4 to low 5 range and I can't explain why my tank would behave differently. There are too many variables when you account for nutrient dosing levels, fauna, CO2 levels, substrate, etc. All I know is that increasing the pH in the tank to match my other tanks, both current and historical, made a positive difference and put it in line with ideal pH targeted by aquasoil manufacturers.
If the tank continues progressing in a positive manner and I have a tank of fully healthy plants, I'll probably do the experiment where I remove the KHCO3 and see if I get stunting/white growth. Single variable experiments are not productive at this moment though because the tank is in rough condition.
@Naturescapes_Rocco I've been doing 70% water changes for a while. I struggled to consistently do 50% because I always remove more water than I want when I'm cleaning up the tank. Since I'm thinking the issue is pH, I'm wondering what yours was at max CO2 with 0dKH vs now. I'm wondering if yours was as low as mine.
It's also possible that a slightly decrease in macros made a difference. I try not to think too hard about these things and just go off of what works. Lower macros+higher KH seems to make a difference so I'm sticking with it for some time.
I would be doing the same thing. If something is working stick with it.If the tank continues progressing in a positive manner and I have a tank of fully healthy plants, I'll probably do the experiment where I remove the KHCO3 and see if I get stunting/white growth. Single variable experiments are not productive at this moment though because the tank is in rough condition.
If things do become really stable that would be the time to start testing theories. The hard part is that it takes a lot of patience. If you were to test the dKH theory, I would lower dKH VERY slowly. Like over a couple of months. Very small changes at one time.
The problem with big changes is that the can issues by the nature of the change while the plants reprogram.
Similar threads
- Replies
- 48
- Views
- 2K

